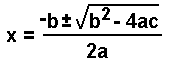
STEP 1: Identify the major species in solution:
( weak acid )
( conjugate base of )
( spectator ion )
( very weak acid or base )
STEP 2: Determine the equilibrium reaction involved:
and will react together to affect the pH of the solution:
STEP 3: Create an I.C.E. grid:
| INITIAL | |||
| CHANGE | |||
| EQUILIBRIUM |
STEP 4: Substitute equilibrium concentrations into the equilibriuum constant expression, then simplify and solve the expression:
Because x 1000 is larger than both 0.10 and 0.12, simplification is not possible. Thus, to reach the solution, it is necessary to use the quadratic formula to solve. Rearrangement gives:
Using the quadratic formula:
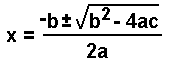

Because we cannot have a negative concentration of ions,
STEP 5: Calculate the pH:
pH = - log = - log( 1.7 x ) = 3.77
How much HCl must be added to 1.00 L of 0.10 M sodium lactate ( ) to make a buffer of the same pH as found in the previous example (3.77)?
STEP 1: Calculate the equilibrium concentration of that will produce the given pH:
x
STEP 2: Determine the equilibrium which must take place for this solution to be a buffer:
For a buffer effect to take place, the solution must contain both the weak acid and its conjugate base. The equilibrium reaction is therefore:
STEP 3: Create an I.C.E. grid:
This time we will call the initial concentration of . Because we know that the equilibrium is so small, we can assume that nearly all the initial is used up.
| INITIAL | |||
| CHANGE | |||
| EQUILIBRIUM |
STEP 4: Substitute the equilibrium concentrations into the equilibrium constant expression:
STEP 5: Solve for :
x
STEP 6: Convert this answer to moles of HCl:
How much NaOH must be added to 1.00 L of 0.10 M lactic acid, to make a buffer of the same pH as the first example (3.77) ?
STEP 1 : Calculate the equilibrium concentration of that will produce the given pH
x
x x x
STEP 2 : Determine the equilibrium which must take place for this solution to be a buffer:
Once again, the weak acid and the conjugate base must be present. This time, however, we want in the equation.
STEP 3: Create an I.C.E. grid
This time we will call the inital concentration of . The given equilibrium should proceed almost to completion, so we will assume nearly all the is used up. Its equilibrium is so small :
| INITIAL | |||
| CHANGE | |||
| EQUILIBRIUM |
STEP 4: Substitute the equilibrium concentrations into the equilibrium constant expression:
This equilibrium is the reverse reaction for a base reacting with water. Therefore, the K we must use is .
STEP 5: Solve for :
STEP 6: Convert this answer to moles of NaOH:
